

Hypha Labs Research Division · USS Olympias Technical Documentation · April 2026
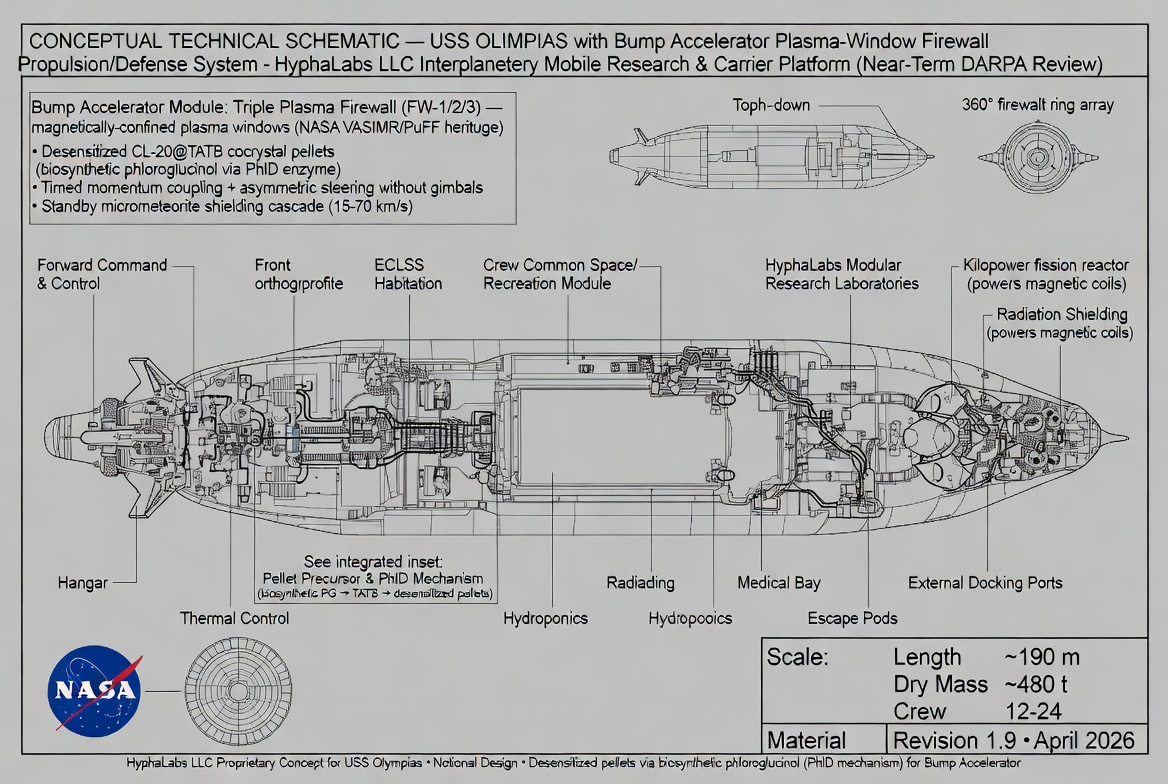
The Bump Accelerator's CL-20@TATB cocrystal pellets require TATB (triaminotrinitrobenzene) as a desensitizing co-crystal partner. Conventional TATB synthesis relies on phloroglucinol (1,3,5-trihydroxybenzene) produced via industrial chlorination chemistry — a process unsuitable for on-mission resupply. Hypha Labs proposes a biosynthetic route to phloroglucinol via the PhlD enzyme (Type III polyketide synthase), converting 3 malonyl-CoA to phloroglucinol in a single-enzyme, chlorine-free reaction (Km ≈ 5.6 μM malonyl-CoA; kcat ≈ 10 min⁻¹). This enables a sustainable pellet resupply chain compatible with long-duration missions where Earth-sourced consumables are unavailable.
The Bump Accelerator is a consumable propulsion system. Every maneuver expends pellets. For short missions — lunar orbit, cislunar transfers — the pellet magazine can be loaded before departure and sufficient propellant carried for the entire mission, just as conventional spacecraft carry their propellant. But the USS Olympias is designed as a long-duration interplanetary carrier. A Mars mission round-trip takes 18–24 months. A deeper solar system mission could take years.
At those timescales, carrying all propellant at departure becomes the dominant mass constraint. Any capability to produce or augment propellant on-mission directly reduces required launch mass — or extends mission range for the same launch mass. The PhlD biosynthetic route addresses this for the TATB component: if malonyl-CoA can be generated from mission feedstocks (food waste, CO₂ atmosphere recycling, onboard carbon-fixing biology), then phloroglucinol can be synthesized onboard, and TATB production follows through known chemical steps.
The biosynthetic route isn't a replacement for pre-loaded pellets on near-term missions. It's the foundation for a resupply architecture that makes very long-duration missions tractable without the tyranny of propellant mass fraction at departure.
CL-20 (hexanitrohexaazaisowurtzitane) has the highest energy density of any conventional explosive currently in production-scale synthesis — approximately 15% higher specific energy than HMX, the previous benchmark. Its cage-like molecular architecture packs more nitrogen-oxygen bonds into a smaller volume than any competitor. The problem is sensitivity: raw CL-20's shock sensitivity is too high for reliable handling in the vibration environment of a spacecraft.
TATB (1,3,5-triamino-2,4,6-trinitrobenzene) is the opposite problem — so insensitive that it is notoriously difficult to reliably initiate. It has been used in U.S. nuclear warhead conventional explosives precisely because it can survive fire, impact, and shock without detonating accidentally. The CL-20@TATB cocrystal combines these extremes at the molecular level: CL-20 provides the energy, TATB's crystal lattice provides the insensitivity. Research by Bolton and Matzger at the University of Michigan (Crystal Growth & Design, 2012) confirmed this cocrystal achieves dramatically reduced sensitivity versus pure CL-20 while retaining ≈80% of its detonation velocity.
Sources: Bolton & Matzger, Crystal Growth & Design 12(9), 4357–4360, 2012. Detonation velocities from NIST/LLNL energetic materials database. CL-20@TATB cocrystal values estimated from published sensitivity reduction data.
Phloroglucinol (1,3,5-trihydroxybenzene) is the industrial precursor to TATB. Conventional synthesis uses phloroglucinol chlorination and amination steps, requiring concentrated chlorine reagents and generating chlorinated waste streams — unsuitable for a closed-loop spacecraft environment.
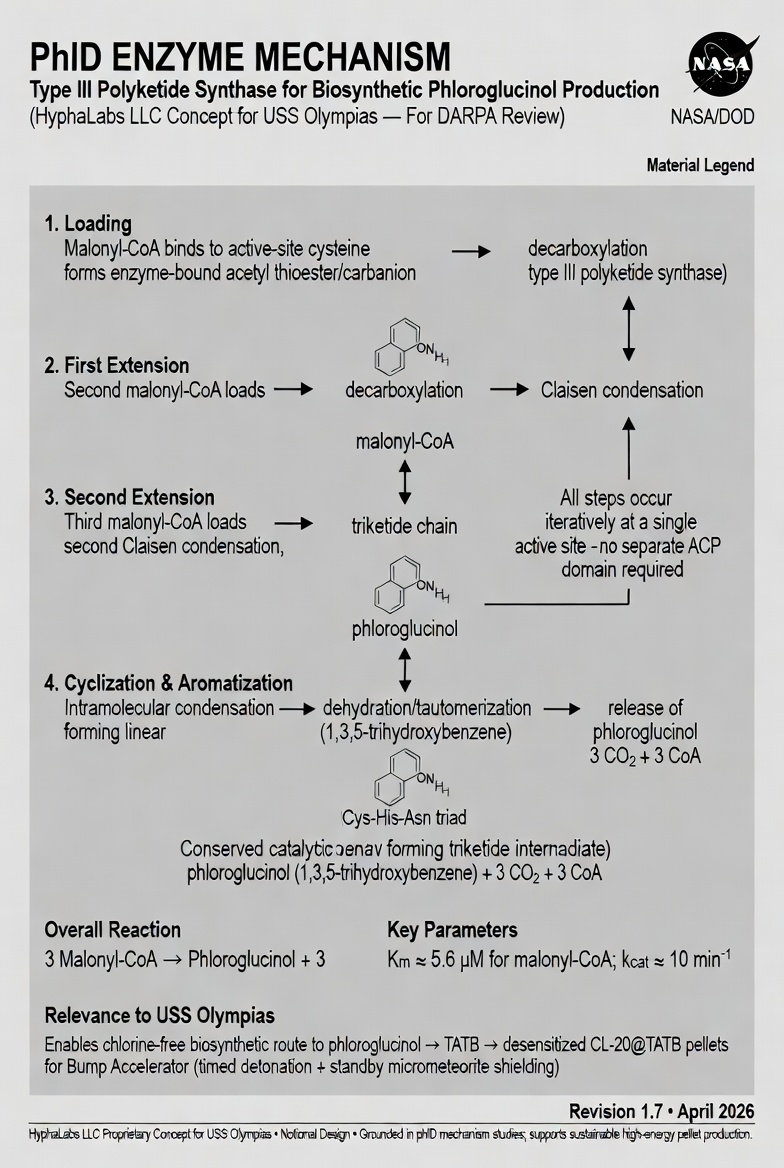
PhlD is a bacterial Type III polyketide synthase originally characterized from Pseudomonas fluorescens, where it produces phloroglucinol as an antifungal compound in the plant rhizosphere. Unlike most polyketide synthases (Type I and II), Type III enzymes are single polypeptides with a single active site that performs all elongation and cyclization steps iteratively — no multi-enzyme assembly line required.
The PhlD reaction is elegantly simple: three malonyl-CoA molecules enter the active site sequentially. The active-site cysteine (Cys-His-Asn catalytic triad) performs iterative decarboxylative Claisen condensation until a triketide intermediate forms, which then undergoes intramolecular cyclization and aromatization to release phloroglucinol, 3 CO₂, and 3 CoA.
The first malonyl-CoA molecule attacks the active-site cysteine residue, forming a covalent thioester bond. Simultaneous decarboxylation of the malonyl group generates an enzyme-bound acetyl carbanion/thioester intermediate. The Cys-His-Asn catalytic triad is conserved from Type I/II PKS and chalcone synthase families — confirming shared evolutionary ancestry across all polyketide synthases.
A second malonyl-CoA binds the active site. Decarboxylation generates a nucleophilic carbanion, which attacks the enzyme-bound thioester via Claisen condensation, extending the chain by two carbons. The diketide-CoA product remains enzyme-bound. All steps occur at the single active site — no acyl carrier protein (ACP) domain transfer required, distinguishing Type III from Type I/II PKS.
The diketide intermediate undergoes a second Claisen condensation with a third malonyl-CoA (following decarboxylation), extending the chain to a triketide intermediate. The triketide remains covalently attached to the active-site cysteine, poised for the final cyclization step. Kinetics: Km ≈ 5.6 μM for malonyl-CoA; kcat ≈ 10 min⁻¹ for the complete 4-step process.
The enzyme-bound triketide undergoes intramolecular aldol condensation (C1-C6 bond formation), forming a 6-membered ring. Sequential dehydration and tautomerization steps drive aromatization to the 1,3,5-trihydroxybenzene (phloroglucinol) product. Hydrolysis releases phloroglucinol from the enzyme, regenerating the active-site cysteine for the next catalytic cycle. Byproducts: 3 CO₂ + 3 CoA (both recyclable within the cell).
3 Malonyl-CoA → Phloroglucinol + 3 CO₂ + 3 CoA
Once phloroglucinol is biosynthesized, the remaining steps to TATB are conventional organic chemistry. Phloroglucinol is first trinitrated to trinitrophloroglucinol (TNPG) using mixed acid nitration, then converted to TATB via ammonolysis — replacement of the three hydroxyl groups with amino groups using ammonium hydroxide or amines under elevated temperature and pressure. This two-step conversion is well-characterized industrial chemistry, but the key breakthrough is that the phloroglucinol feedstock no longer requires chlorinated precursors.
Conventional phloroglucinol synthesis starts from benzene-1,3,5-triol produced via chlorination and reductive dechlorination sequences — industrial steps that require bulk chlorine handling. The PhlD route generates the identical starting material from malonyl-CoA via a clean enzymatic pathway, with no chlorinated intermediates.
The PhlD route only closes the sustainability loop if malonyl-CoA itself can be generated onboard from mission-available feedstocks. Malonyl-CoA is a central metabolite in fatty acid synthesis — present in essentially all living cells that consume acetyl-CoA from carbohydrate catabolism. On USS Olympias, three potential malonyl-CoA sources exist:
The complete biological-to-propulsion loop is not near-term — it requires integration of multiple biological systems at production scale within a spacecraft mass budget. But the individual components (PhlD characterization, ACC overexpression, mycelium bioreactor operation) are all laboratory-validated. The synthesis challenge is engineering, not fundamental science.
Hypha Labs Research Division · USS Olympias Technical Documentation · April 2026
For technical inquiries, program manager briefings, or collaboration — contact us directly.